
CARE TRIAL: LATEST NEWS!
Care Study site teams across the country have successfully recruited 57 participants for the first ever pilot #clinicaltrial for #cavernoma. There are now only 3 participants left to recruit before the end of April.

Care Study site teams across the country have successfully recruited 57 participants for the first ever pilot #clinicaltrial for #cavernoma. There are now only 3 participants left to recruit before the end of April.

We are delighted to announce that a consortium, led by CAUK medical advisor Professor Rustam Al-Shahi Salman and comprising of a number of leading clinicians and patient advocacy organisation leaders – including David White of CAUK – has been awarded a grant to initiate the planning of an international ‘platform trial’ to test multiple potential cavernoma medications at a time.

We are delighted to report that the team at Kings College Hospital London has recently recruited its first two participants for the trial, with one patient randomised for ‘active treatment’ and the other randomised for ‘conservative management’. In equally exciting news, the team at Royal Hallamshire Hospital in Sheffield have recruited their sixth participant!
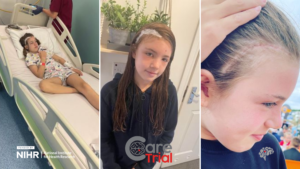
Elise was diagnosed with cavernomas at just 1 year old, a year a
fter her mother Suzan’s initial diagnosis. After learning to walk and talk, and achieving the typical developmental milestones you might expect of any infant, out of nowhere Elise began to regress. She had suddenly lost all of her acquired skills and Suzan noticed that she was frequently holding her head and screaming in pain.
We’re pleased to announce that the CARE Study is almost ready to open to recruitment! We will be opening our first recruitment centre in July.

Join our science advisor David White in a new part of his Science of Cavernoma Series, as he discusses how Cavernoma Grow : The role

Researchers have discovered an explanation for why cavernoma – clusters of dilated blood vessels in the brain – can suddenly grow to cause seizures or stroke.

The Cavernomas A Randomised Effectiveness (CARE) pilot trial CAUK is the Patient Group in a consortium also including senior clinicians with a special interest in

The influence of the microbiome (the bacteria in your gut) on cavernoma development N.b. In the US, cavernoma are called cavernous angioma Introductory text for
PO Box 366
Watlington
Oxfordshire
OX10 1GF
Registered Charity 1114145
Scottish Charity SC048458
Join CAUK today to gain access to our support services and free events